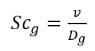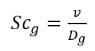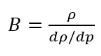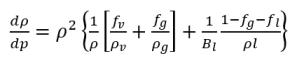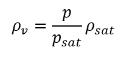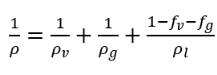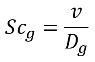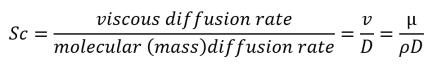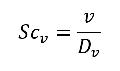Definitions
• Aeration—Component of the cavitation model that accounts for the NCG in a liquid system.
• All Gas Volume—Combined volume of the free NCG, dissolved gas, and vapor in a liquid for a selected volume.
• All Gas Volume Fraction or All Gas Vol Fraction—Average volume fraction of the free NCG, dissolved gas, and vapor in a liquid for a selected volume.
• Dissolved Gas—NCG dissolved in the liquid. It is included in the equilibrium dissolve gas model, dissolved gas model, and full gas model.
• Dissolved Gas Schmidt Number—Schmidt number of the dissolved NCG in a liquid. It is used to compute the mass diffusivity in the dissolved gas model.
where,
Dg,d | mass diffusivity of the NCG through the liquid (m2/s) |
• Dissolved Gas Volume—Total volume of the dissolved NCG in a liquid for a selected volume. The density of the dissolved gas is made up to be the same as the liquid density.
• Dissolved Gas Volume Fraction—Volume fraction of the dissolved NCG in a liquid for a selected volume.
• Dissolved Mass Fraction—Mass fraction of the dissolved NCG in a liquid at a given location.
• Effective Bulk Modulus with Cavitation—For cavitating flows, the effective bulk modulus B is based on the mixture of the liquid and gas. Following the Nykanen-model (Nukanen 2000), B is defined in terms of the mixture density (ρ) of liquid, vapor, and noncondensable gases:
equation 2.158
and
equation 2.159
where,
l | liquid |
v | vapor |
g | all the noncondensable free gases in the system |
ƒ | mass fraction |
ρ | local flow pressure |
Bl | liquid bulk modulus |
The mixture density is also referred to as effective fluid density defined in
equation 2.161.
• Effective Vapor Density—Without the consideration of the temperature effect, the local (effective) vapor density is calculated using Boyle’s Law:
equation 2.160
where,
psat | saturation pressure |
ρsat | density at the given temperature |
If the temperature varies because the Heat module is active, the vapor density at saturation is also a function of temperature. Use the Expression Editor to include the thermal effects.
• Effective Viscosity—Viscosity of the fluid mixture in the domain. Creo Flow Analysis, by default, does not include the effects of vapor or NCG on the mixture viscosity (liquid viscosity only).
• Fluid Density (Effective) with Cavitation—Calculated based on the respective mass fractions and densities of the liquid, fluid vapor, and NCG such as air according to the following formula:
equation 2.161
where ρv, ρg, ρl, ρv follow their respective laws of density; and ƒv, ƒg, ƒl are directly specified or obtained from solving transport equations, depending on the components and cavitation models.
• Gas Mass Fraction—Mass fraction of the nondissolved (free) NCG in the fluid, such as mass fraction of free air in liquid water. It is also referred to as Free Gas Mass Fraction, which does not include any dissolved noncondensable gas.
• Gas Schmidt Number—Schmidt number of the NCG in a liquid. It is used to compute the mass diffusivity D in the variable gas mass fraction model and full gas model.
where,
v | kinematic viscosity of the primary fluid (m2/s) |
Dg | mass diffusivity of the NCG through the liquid (m2/s) |
• Gas Volume—Total volume of the free NCG in a liquid for selected volumes.
• Gas Volume Fraction or Gas Vol Fraction—Volume fraction of the free NCG in a liquid for selected volumes.
• Liquid Volume—Total volume of the liquid for selected volumes.
• Liquid Volume Fraction—Volume fraction of the liquid for selected volumes.
• NCG or Noncondensable Gas—Gas in the liquid that is different from the vapor of the liquid. As such, it can dissolve or be released, but is not generated or absorbed by evaporation or condensation. The density is computed based on the gas molecular weight, the local pressure and temperature, according to the ideal gas law.
• Schmidt Number—Defined as:
equation 2.162
where,
ρ | density (kg/m3) |
v | kinematic viscosity (m2/s) |
D | mass diffusivity (m2/s) |
µ | dynamic viscosity (Pa-s) |
• Total Volume Fraction—Local volume fraction of the combination of free NCG, dissolved gas, and vapor relative to the total fluid volume.
• Vapor—Gas phase of a liquid substance at a temperature lower than its critical temperature, such as water vapor.
• Vapor Mass Fraction—Total mass fraction of vapor in the fluid.
• Vapor Schmidt Number—Schmidt number of the vapor in the liquid. It is used to calculate the mass diffusivity Dvapor of the vapor in all cavitation models.
where,
Dv | mass diffusivity of the vapor in through the primary liquid (m2/s) |
• Vapor Volume—Total volume of the vapor in a liquid for a selected volume.
• Vapor Volume Fraction—Volume fraction of the vapor. In the add XY-plot menu it is abbreviated as Vapor Vol Fraction.
• Volume Fraction—Volume of component divided by the total volume within a selected volume or at a point.